- Best Seller
IRON FORTE
Based on 6 reviews
1 capsule per day with a glass of water with a meal. For adults only.
The benefits
-
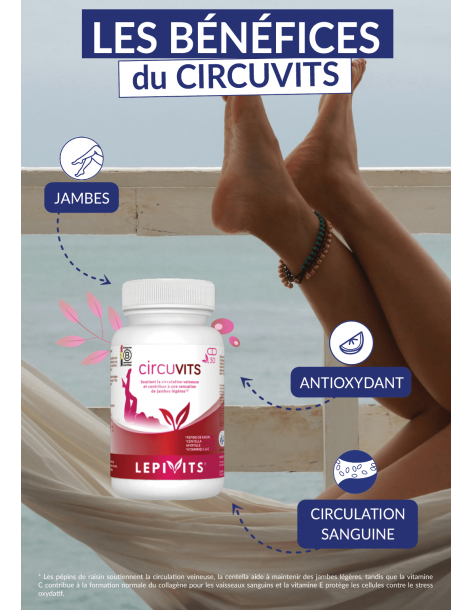
 Immunity
Iron and vitamins B6 and B9 contribute to the proper functioning of the immune system.
Immunity
Iron and vitamins B6 and B9 contribute to the proper functioning of the immune system.
-
 Energy
Iron and vitamins B6 and B9 help to reduce fatigue. Vitamin B6 contributes to the normal functioning of the nervous system.
Energy
Iron and vitamins B6 and B9 help to reduce fatigue. Vitamin B6 contributes to the normal functioning of the nervous system.
-
 Red blood cells
Iron and vitamin B6 contribute to the normal formation of red blood cells. Iron supports the formation of haemoglobin. Folate contributes to the normal formation of blood.
Red blood cells
Iron and vitamin B6 contribute to the normal formation of red blood cells. Iron supports the formation of haemoglobin. Folate contributes to the normal formation of blood.
Product qualities
-
 Pullulan capsule vegetable
Pullulan capsule vegetable
-
 No preservatives, no pesticides, no artificial colours or flavours
No preservatives, no pesticides, no artificial colours or flavours
-
 Vegan
Vegan
-
 Recycling
Recycling
-
 Quatrefolic
Quatrefolic
Nutritional information
| NUTRITIONAL INFORMATION | 1 CAPSULE | AR%* |
|
Iron element Vitamin B6 Vitamin B9 |
45mg 6mg 300μg |
321 428 150 |
* Recommended Daily Allowance (Directive 2008/100/EC amending -> Decree 08.01.1992)
Optimize your vitality with Fer Forte
Fer Forte is a dietary supplement designed to meet the iron needs of individuals with specific diets or requirements, such as pregnant women, vegetarians, or vegans. This product utilizes a bioavailable iron salt, iron bisglycinate, known for its excellent absorption and superior digestive tolerance.[1] Enriched with vitamins B6 and B9 in their bioactive forms, Fer Forte offers an effective synergy to support overall health.
Fer Forte is formulated to provide an optimized source of iron, an essential element for the proper functioning of many biological processes. Iron is vital as it promotes the formation of hemoglobin, the protein in red blood cells that transports oxygen from the lungs to all parts of the body.[2] This transport function is important not only for maintaining our energy levels and vitality but also for supporting the proper functioning of the immune system.
The body requires iron for several essential functions. It may not produce enough healthy red blood cells, and without an adequate amount of iron, this can lead to chronic fatigue, weakness, and a weakened immune system.
Maximize iron absorption with Fer Forte
- Conditionnement
- 60 capsules
- Composition
- Iron bisglycinate, capsule shell: pullulan, rice starch, vitamin B6 (pyridox- al- 5'-phosphate), (6S)-5-methyltetrahydrofolic acid in the form of a glucosamine salt (Quatrefolic®).
- Main indication
- Fatigue
- Specific indication
- Iron deficiency anaemia
- Dosage
- 1 capsule per day with a glass of water with a meal. For adults only.
- Recommandations
- This food supplement is not a substitute for a varied and balanced diet and a healthy lifestyle and is not a medicinal product. Inform your doctor and/or pharmacist if you are taking any medication at the same time. Product for adults only. Not suitable for children, adolescents. Keep out of reach of children. Store at room temperature and in a dry place. Do not exceed the recommended daily dose.
- Notification number
- 3760/20
- Posologie (nbr gélules/jour)
- 1
- Condition
- New product
- ean13
- 5430002936673